Fritz Haber: Chemist, Nobel Laureate, German, Jew. New York, NY: Springer-Verlag, 1969.īorn, Max. My Life: Recollections of a Nobel Laureate. 9.1, "Predicting the Geometry of Molecules and Polyatomic Ions."īorn, Max. 8.5, "Lewis Structures and Covalent Bonding." 8.3, "Lattice Energies in Ionic Solids." Equal bond energies imply equal spatial disposition, and the electronic structure dictates bond disposition, which dictates molecular architecture. This lecture also introduces the valence shell electron pair repulsion (VSEPR) model, properties of covalent (saturated, directional) and ionic bonds, rules for determining molecular shapes, and the classification of each electron as a bonding electron (B) or a nonbonding electron (NB).
#Do born haber cycle problems download
> Download English-US caption (SRT) Resources
#Do born haber cycle problems archive
> Download from Internet Archive (MP4 - 214MB)

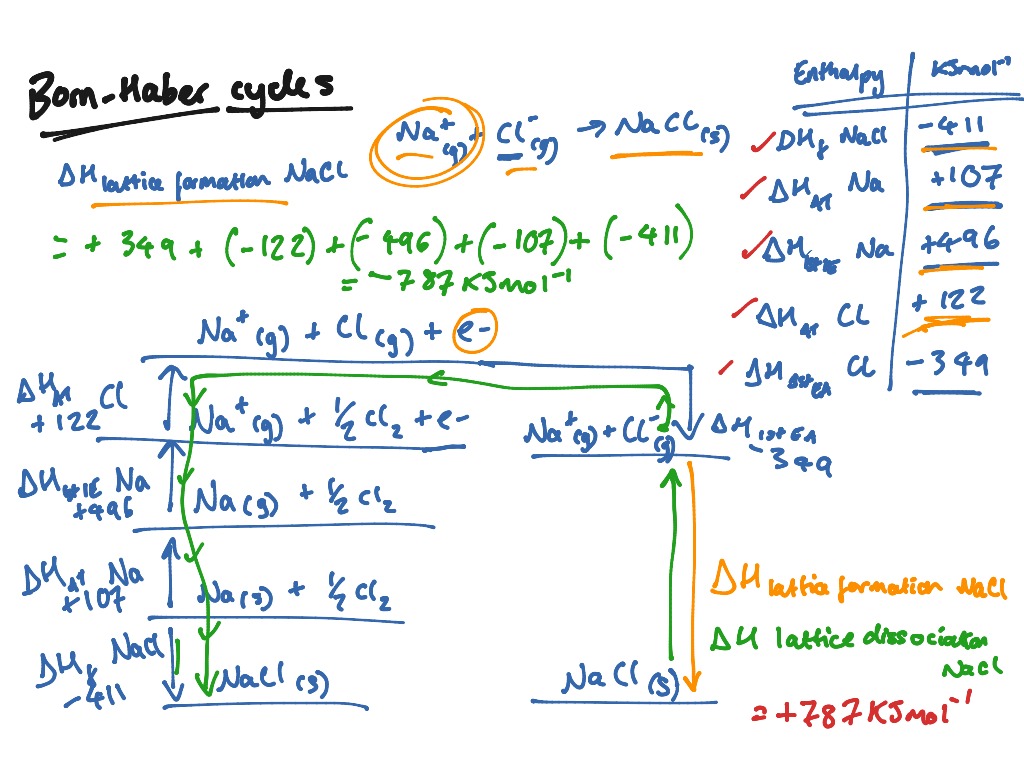
List the properties of ionic crystals, and relate them to the lattice energy.Sketch the potential energy as a function of inter-ionic separation.Understand the valence shell electron pair repulsion (VSEPR) model.Describe quantitatively the energetic factors and characteristics involved in the formation of an ionic bond.Learning ObjectivesĪfter completing this session, you should be able to: Sadoway discusses the shortcomings of ionic bonding and Lewis's concept of shell filling by electron sharing including the Lewis dot notation ( Session 9) and returns to the valence shell electron pair repulsion (VSEPR) model in Session 11: The Shapes of Molecules. Octet stability and what it means in terms of shell filling ionic bonding and its formation as a result of Coulombic attraction between a cation and an anion ( Session 7).Sodium chloride (NaCl), manganese (Mn), sodium (Na), potassium (K), silver iodide (AgI), neon (Ne), magnesium oxide (MgO), aluminum (Al), aluminum oxide (Al 2O 3), cryolite (Na 3AlF 6)ĭesign of thermal abrasion resistance materials, design of inert anode materialsīefore starting this session, you should be familiar with: ion lattice, and properties of ionic crystals, enthalpy of reaction: Hess's law, Born-Haber cycleīorn exponent, cation, anion, covalent bond, ionic bond, valence shell electron pair repulsion model (VSEPR), interionic separation, crystal array, omnidirectional bond, unsaturated bond, Avogadro's number, electrostatic energy, ionic solid, Madelung constant, melting point, boiling point, electrical insulator, hardness, brittle, soluble, polar solvent, noble gas, ionic liquid, photon, transparency, binding energy, hybridized bond, elasticity, enthalpy, ionization energy, sublimation, electron affinity, lattice energy, bonding electron, nonbonding electron, molecular architecture Ionic bonding: octet stability by electron sharing, energy of ion pairs vs.
